SaaS Solution · Pharmaceutical Industry
SaaS Solution · Pharmaceutical IndustryEffortless Cleaning validation,
no stress - no Excel
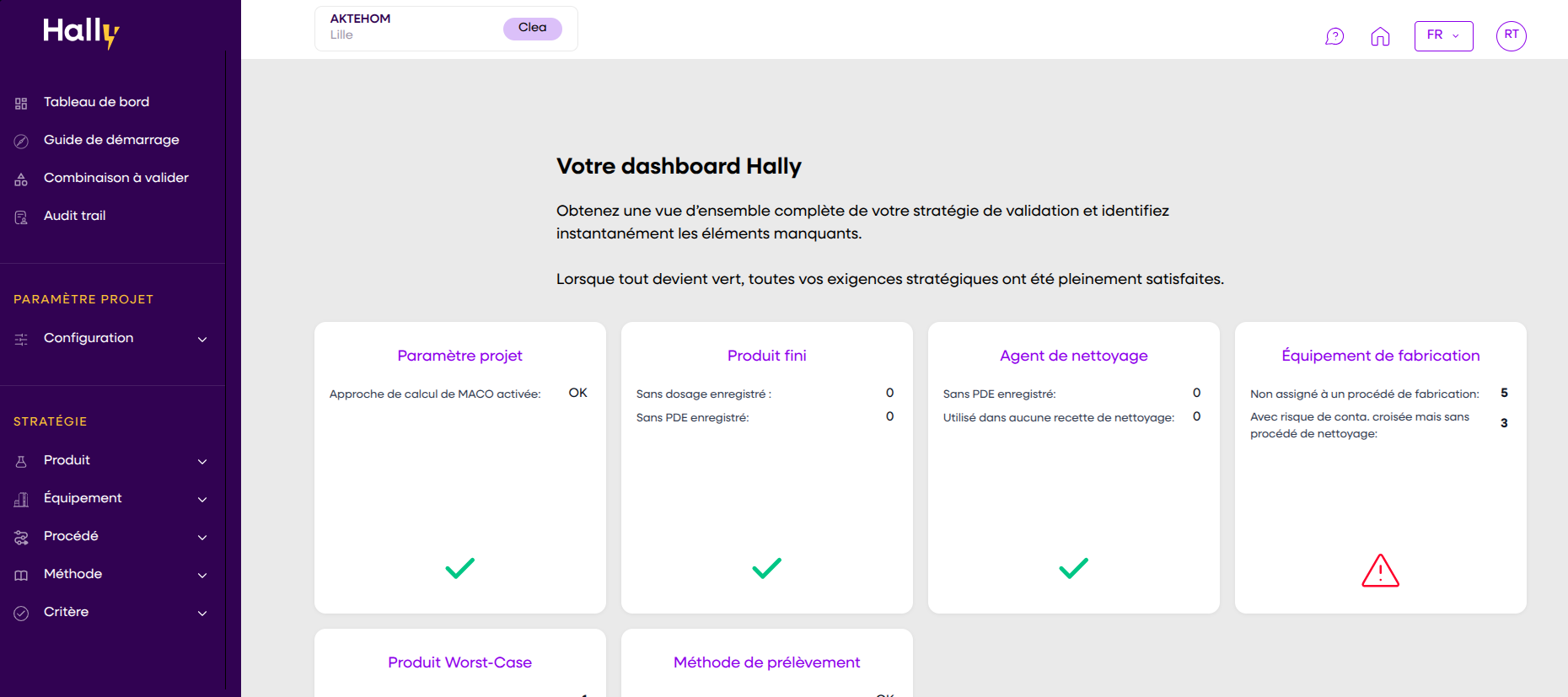
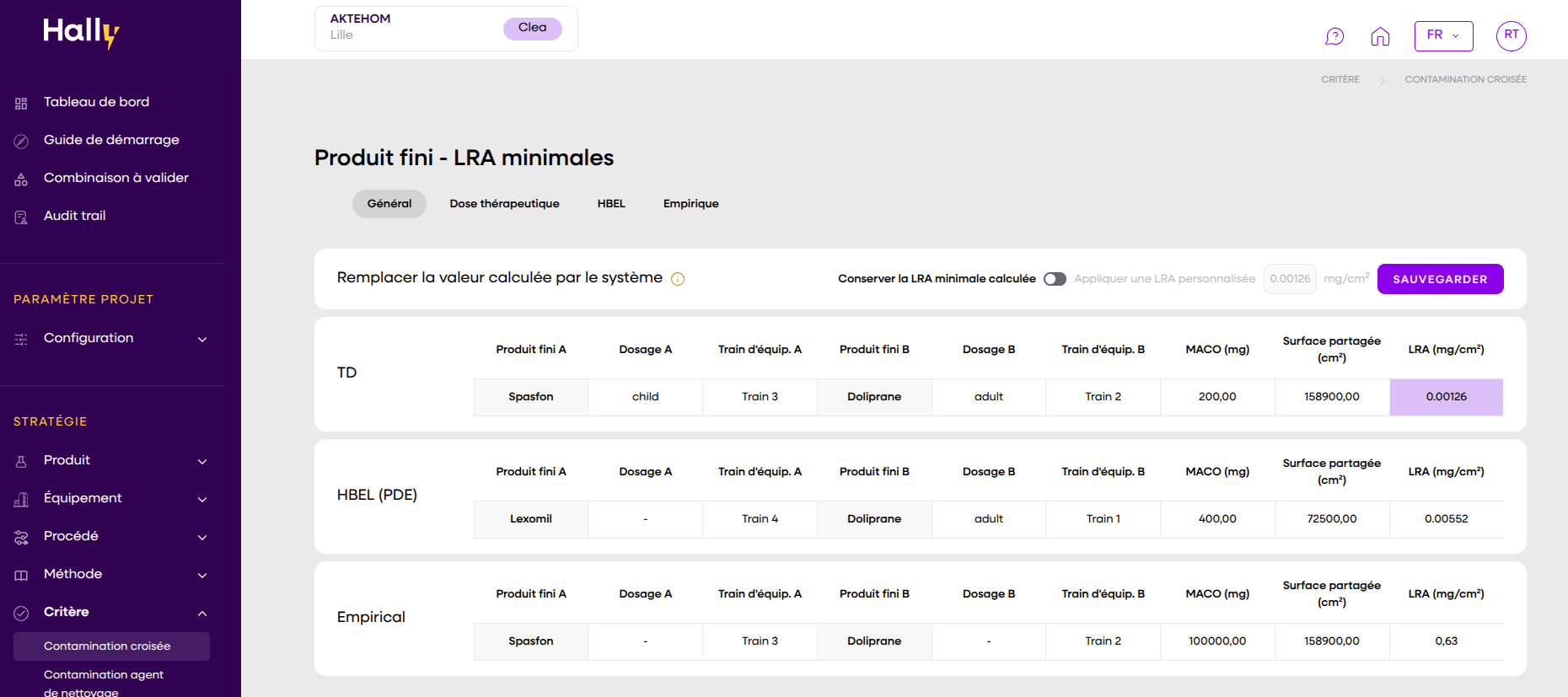
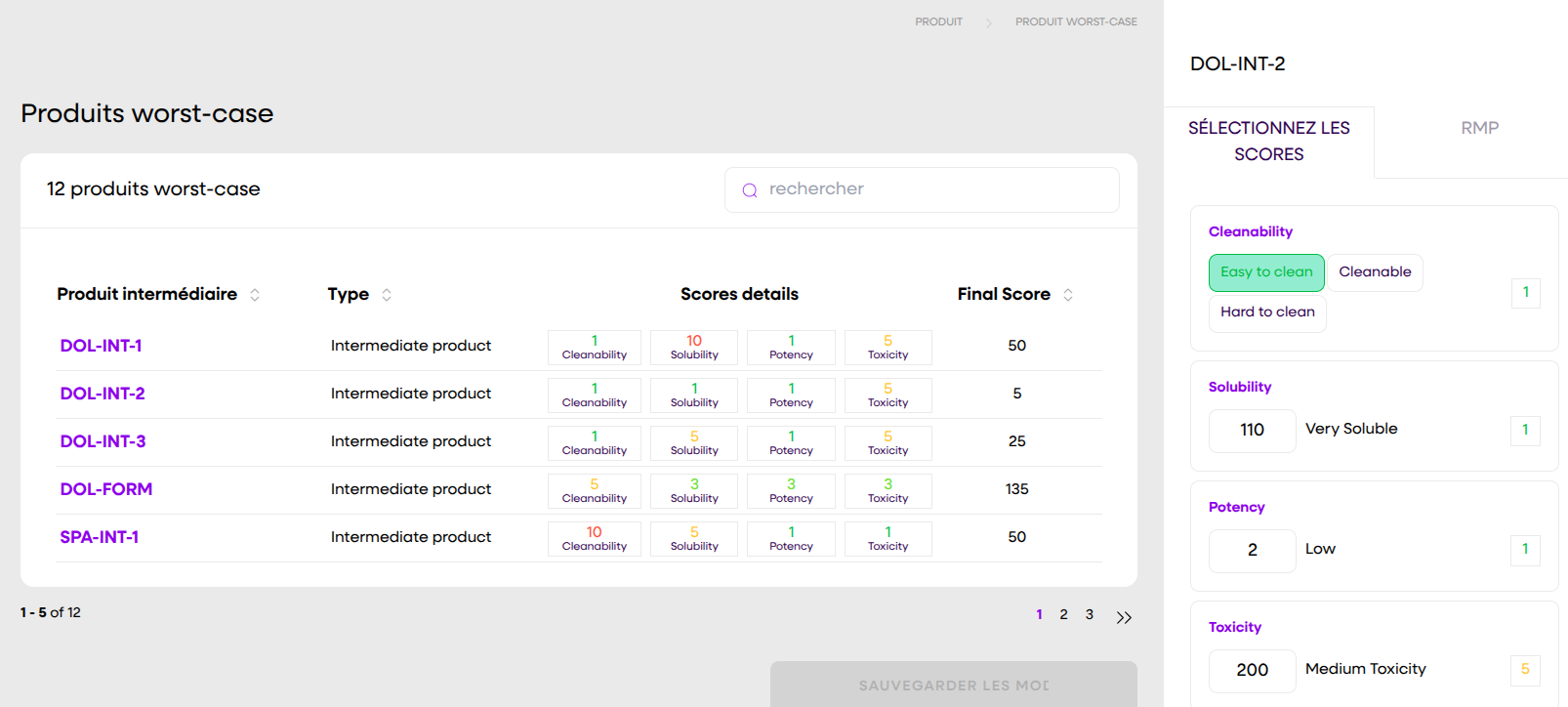
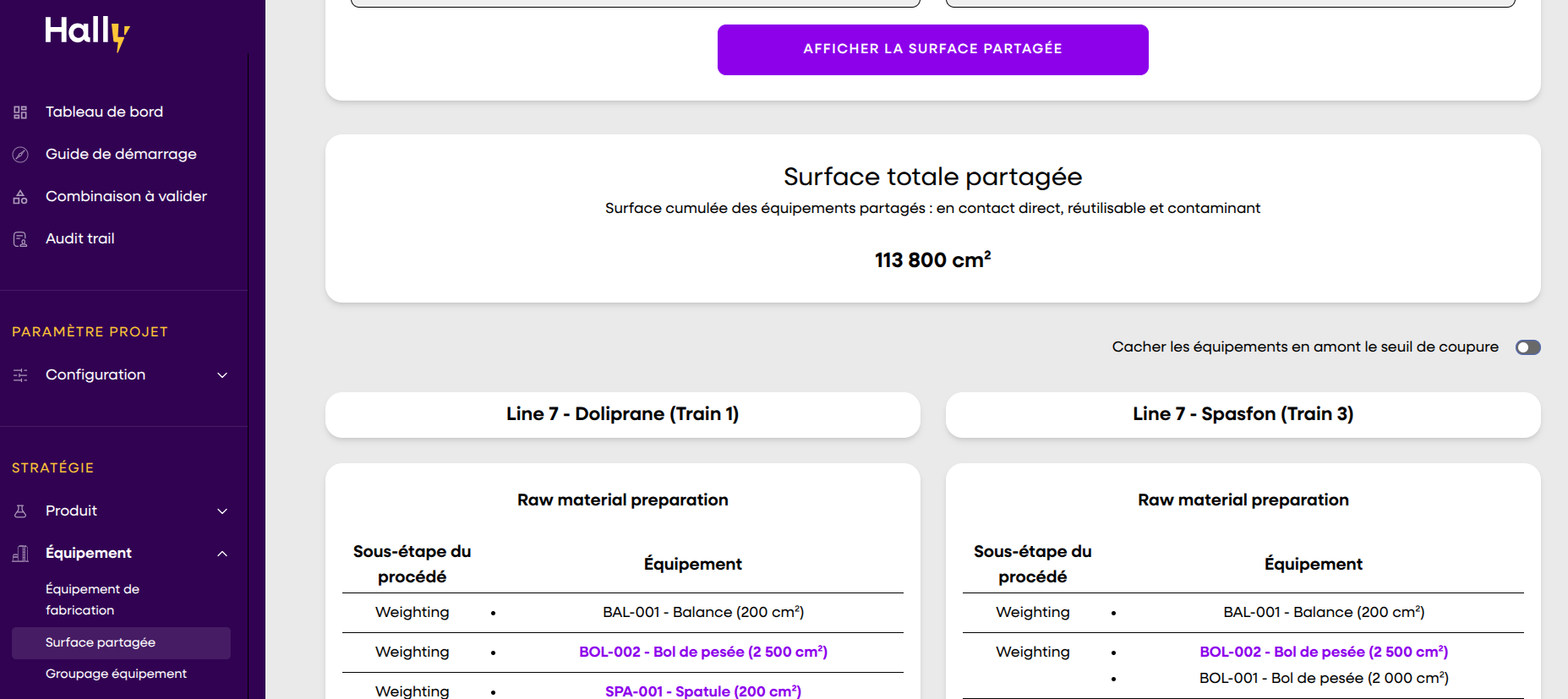
In an industry where everything is complex, a clear and effective solution makes all the difference. Automate your strategies, centralize your data, and pass inspections with confidence.
✓ No credit card · ✓ Demo space included · ✓ Immediate access